g. Methicillin-Resistant Staphylococcus aureus
|
|
Approximately 32% (89.4 million persons) and 0.8% (2.3 million persons) of the US population are colonized with S aureus and MRSA respectively (Kuehnert et al. 2006). The number of healthcare-associated staphylococcal infections due to MRSA is on the rise—from 2% of S aureus infections in US intensive care units (ICUs) in 1974 to 22% in 1995 and 64% in 2004 (Klevens et al. 2006). The rate of MRSA has been found to be as high as 59.2% in hospital wards (Naber 2009). There are an estimated 292,000 hospitalizations due to S aureus infections annually in US hospitals (Kuehnert et al. 2005). Of these, approximately 126,000 hospitalizations are related to MRSA. Nosocomial MRSA infections with reduced susceptibility to vancomycin are found to be more virulent than other strains and are a major contributor to in-hospital morbidity and mortality (The Medical Letter 2009; Peleg et al. 2009; Xiong et al. 2009).
In 2007, rates of MRSA bacteremia ranged from 0.8% in Denmark to 52.4% in Malta (Naber 2009). In Belgium, the MRSA bacteremia–associated mortality among critically ill patients was 23.4% compared with 1.3% for methicillin-susceptible S aureus (MSSA) strains (Blot et al. 2002). The estimated risk of mortality associated with MRSA bacteremia compared with MSSA bacteremia is 1.93 (Cosgrove et al. 2003) with a relative risk of 2.12 (Whitby et al. 2001).
In Europe, S aureus, including MRSA, are the leading isolates in skin and soft tissue infections (SSTI), with a range of 10% to 51% (Kollef et al. 2008). A study of 9 European country hospitals revealed that 28% of patients with bacteremia received less than optimal therapy, resulting in higher morbidity and mortality rates (Ammerlaan et al. 2009). A study of MRSA and MSSA from 450 European hospitals in 26 different countries showed that strains of MRSA tend to cluster in individual healthcare facilities and also within regional borders. This suggests the spread of MRSA through multiple admissions to different care facilities by infected patients (Grundmann et al. 2010). Spatial mapping and geographic information could help identify the patterns of spread and help in the control of the MRSA transmission (Lowy 2010).
Figure 1. Prevalence (%) of MRSA Among EU Patients Presenting With Bacteremia
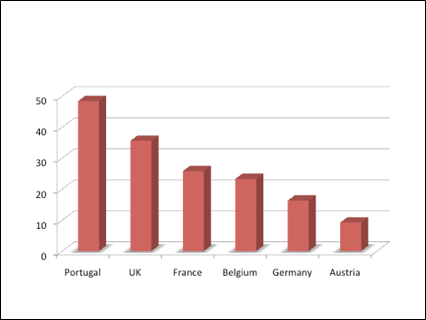
Adapted from: Naber CK. Staphylococcus aureus bacteremia: epidemiology, pathophysiology and management strategies. Clin Infect Dis. 2009;48(suppl 4):231-237.
It is estimated that more than 20% of all HCWs are carriers of MRSA (Chan et al. 2009). Of these, 88% are nasal carriers with the remainder being throat carriers. Colonization with MRSA can persist for to 2 years or longer (Robicsek et al. 2009). Aerosols containing MRSA are commonly found in many residential homes (Gandara et al. 2006).
MRSA is a significant cause of infections outside of the hospital, too (Johnson et al. 2009). Community-acquired MRSA (CA-MRSA) strains have increased dramatically during the last 2 decades (Klevens et al. 2007). It is estimated that 13.7% of invasive MRSA infections are community acquired (Klevens et al. 2007). Between 6% and 21.7 % of end-stage renal disease patients are colonized with MRSA (Wang et al. 2009; Johnson et al. 2009), and the prevalence of CA-MRSA colonization increases with the number of years that a patient is on dialysis (Lin et al. 2009). Available evidence suggests that CA-MRSA pneumonia continues to be associated with seasonal influenza virus, including the 2009 A (H1N1) pandemic (Murray et al. 2010). The Infectious Diseases Society of America (IDSA) recently issued guidelines for prevention and treatment of MRSA (Liu et al. 2011).
Risk factors for CA-MRSA include:
-
Group living and activities that increase the frequency of personal physical contact
-
Poor personal hygiene practices and inadequate hand washing.
-
Scarce resources or inappropriate hygiene practices among group living (such as dormitories, military barracks, prisons, and locker rooms) and activity settings (such as physical impact sports)
-
Insufficient local surveillance system and laboratory diagnostic capacity
-
Inappropriate use of antibiotics
Beyond the conventional designation of nosocomial and community strains, novel MRSA strains have now been identified worldwide in domesticated animals (particularly swine), which are also capable of transmitting infections to humans (Smith and Pearson 2011). A MRSA mecA (LGA251) was recently isolated in United Kingdom and Denmark laboratories that require special diagnostic testing for detection (García-Álvarez et al. 2011).
|




